.jpg?x=1164&format=.jpg)

 Favorite (5)
Favorite (5)
Ebnet Medical GmbH from Schwerin, Germany, develops innovative devices for vascular access. An innovative Peripheral Intravenous Catheter (PIVC) is designed for future reduction of frequent, severe and costly complications. The founder, a specialist in anesthesiology, intensive care and emergency medicine, is still an active emergency physician.
The innovative tool is designed for future reduction of frequent, severe and costly complications. It can be easily integrated in existent workflows. Improved PIVC control and more precise insertion into the vein by only one hand will increase first puncture success rates. Ultrasound guidance can be continuously applied by the other hand. In contrast to product solutions currently marketed or under development, a guide wire will not be necessary to create superior performance and hence clinical value. Simplicity and safety in vascular access are key paradigms of Ebnet Medical when finalizing product development. The aim is to develop medical products without increasing technical or product complexity. Application and material costs as well as waste will be reduced to a minimum. Supply chains will be kept short. Ebnet Medical systematically addresses relevant milestones to decrease development and investment risks.
A positive correlation between Ebnet PIVC and beneficial outcomes has to be proven in studies. Evidence generation for a superior functionality and performance was initiated even at an early stage. A new PIVC prototype series (Minimum Viable Products) was evaluated in suitable test environments (not in patients) by clinical experts at a renowned German University Hospital. In a first step, the innovative functionality was confirmed. The product will also address catheter tube kinking and blockage. In daily medical routine, kinking of catheter tubes and infusion lines leading to costly complications and reduced period of use still occurs quite frequently.
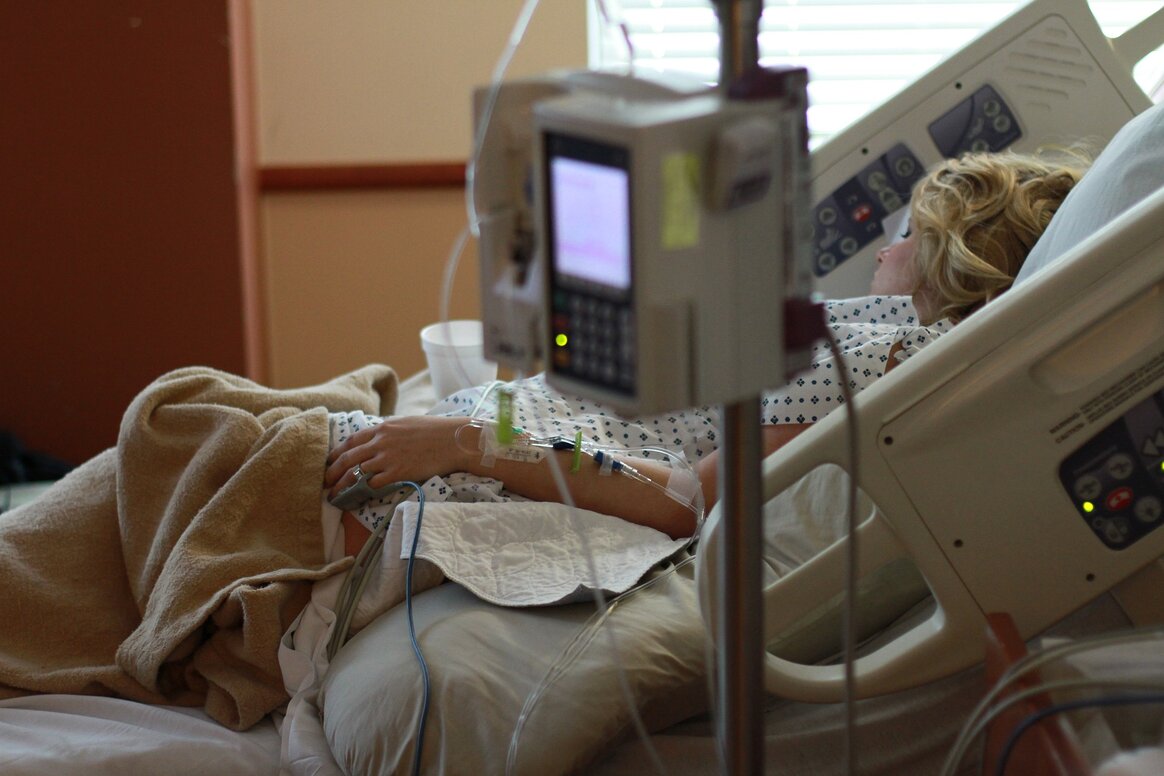
Kinking can be dangerous for patients, e.g. when the flow of an anesthetic/sedating agent (e.g. during surgery) or of a life-saving therapeutic (e.g. chemotherapeutic) is interrupted during the medical procedure. Kinking catheter tubes and infusion lines should not be accepted. A new technology under development by Ebnet Medical offers improved solutions for a non-kinking design of catheter tubes and infusion lines. Successful first tests of non-kinking catheter tube prototypes were already run. Positive feedback from clinical experts was obtained. The aim is to ensure catheter tube patency to increase PIVC lifespan and cost-effectiveness significantly. The new technology is suitable for all types and diameters of catheter tubes and infusion lines. It is, beside a core patent application, extensively secured by other Ebnet Medical patent applications. The company also filed patent applications for PIVC components to make fixation of PIVC to the skin faster and more reliable. Catheter dislodgement is a major concern for patients and medical experts.
The Ebnet PIVC is therefore designed to overcome most relevant weaknesses of conventional PIVCs in the future.
Due to its need- and value-driven business approach, Ebnet Medical has a deep understanding of links between medical practice and technical innovation and offers practical solutions. Feedback from clinical experts is continuously obtained. Further feedback is highly appreciated and can be directly considered for product development.
Its extensive and constantly growing international patent portfolio allows Ebnet Medical to create a competitive advantage in vascular access innovation. Safety in vascular access leading to excellent patient care is our top concern. Certification pathways and timelines for EU and US are evaluated simultaneously with the aim to shorten time to market introduction. Modifications and follow-on products will lead to a disruptive and scalable platform technology for various catheter types in human and veterinary medicine.
.jpg?format=.jpg&upscale=False&x=516)
Ebnet Medical GmbH, Schwerin, Germany